|
1/13/2024 0 Comments Viscosity of water at 300k 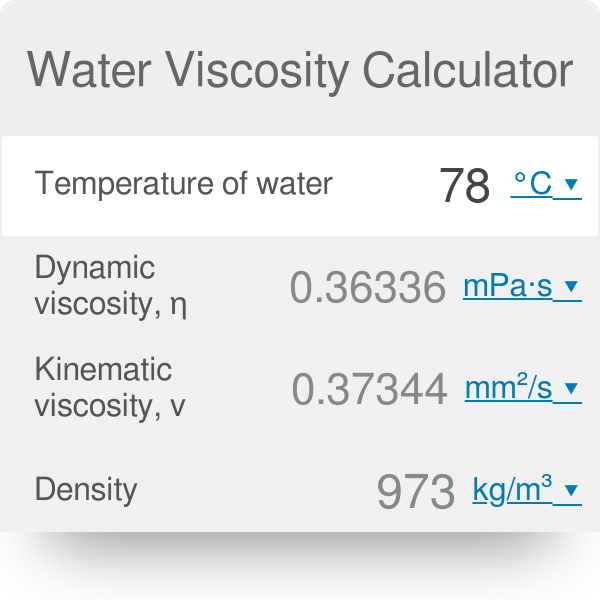 
Probing with viscosity the effect of pressure on the hydrogen bond network can shed light on the debate about the origin of water anomalies.Īnother puzzle associated with the dynamic properties of water is its fragile-to-strong crossover ( 21). Huge efforts have thus been made to improve our knowledge of supercooled water see, for instance, the recent achievements about structural ( 18), thermodynamic ( 19), and dynamic ( 11, 20) properties. Properties of supercooled water are a key to explain water anomalies, and to choose between several proposed theories ( 16, 17). The increase of the viscosity anomaly can only be inferred from interpolation between ambient pressure data for supercooled water ( 10, 11) and high pressure data for stable water ( 12, 14, 15). However, for viscosity, whereas extensive data are available for stable water ( 9) and for supercooled water at ambient pressure ( 10, 11), there is none in supercooled water under pressure, except for one single data point only slightly supercooled ( 12). Similar anomalies exist in the stable liquid for other dynamic properties, and have been found to become more pronounced when the liquid is supercooled (i.e., in the region beyond the melting curve) for self-diffusion ( 5, 6) and rotation ( 7, 8). Viscosity increases with applied pressure for all liquids, except for water below 306K as first noticed in 1881 ( 1 – 4). Our results support the idea that water is a mixture of a high density, “fragile” liquid, and a low density, “strong” liquid, the varying proportion of which explains the anomalies and fragile-to-strong crossover in water. Inspired by a previous attempt, we show that a remarkably simple extension of a two-state model, initially developed to reproduce thermodynamic properties, is able to accurately describe dynamic properties (viscosity, self-diffusion coefficient, and rotational correlation time) as well. Here we report measurements with a time-of-flight viscometer down to 244K and up to 300MPa, revealing a reduction of viscosity by pressure by as much as 42%. However, surprisingly, no data are available for the viscosity of supercooled water under pressure, in which dramatic anomalies are expected based on interpolation between ambient pressure data for supercooled water and high pressure data for stable water. At low temperature, water at ambient pressure becomes more tetrahedral and the effect of pressure becomes stronger. Viscosity is thus a macroscopic witness of the progressive breaking of the tetrahedral hydrogen bond network that makes water so peculiar. It occurs concurrently with major structural changes: The second coordination shell around a molecule collapses onto the first shell. The anomalous decrease of the viscosity of water with applied pressure has been known for over a century. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
